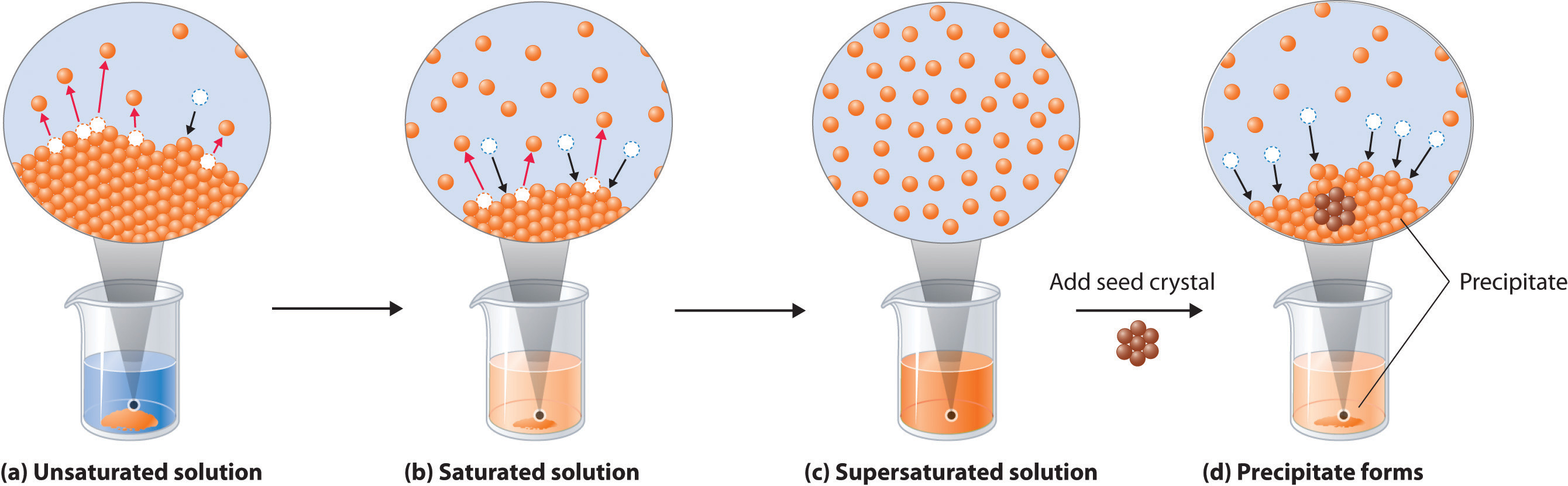
SOLVED: Solubility (Very Soluble; Soluble; Partially Soluble, Insoluble) Lipid Water Acetic Anhydride Acetone Petroleum Ether Hexane Oleic Acid Vegetable Olive Oil Butter Detergent

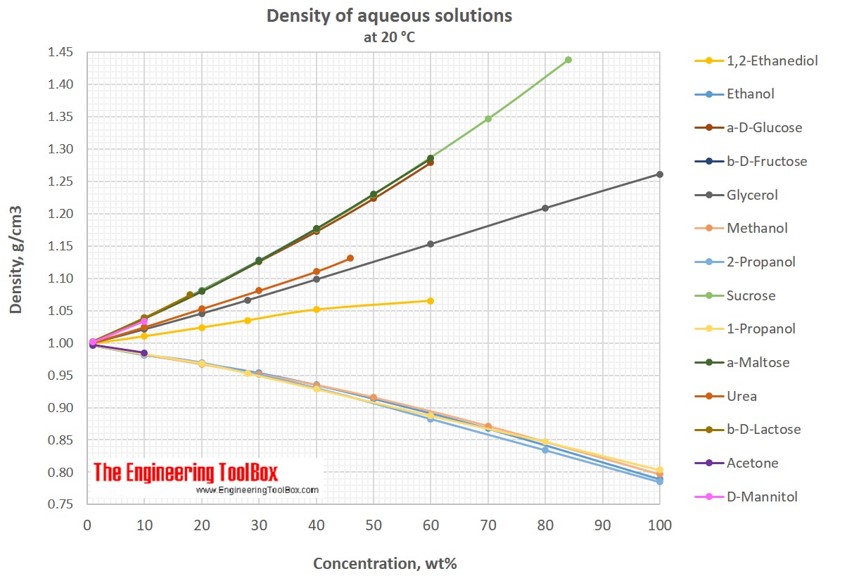
Solubility of d-Glucose in Water and Ethanol/Water Mixtures | Journal of Chemical & Engineering Data
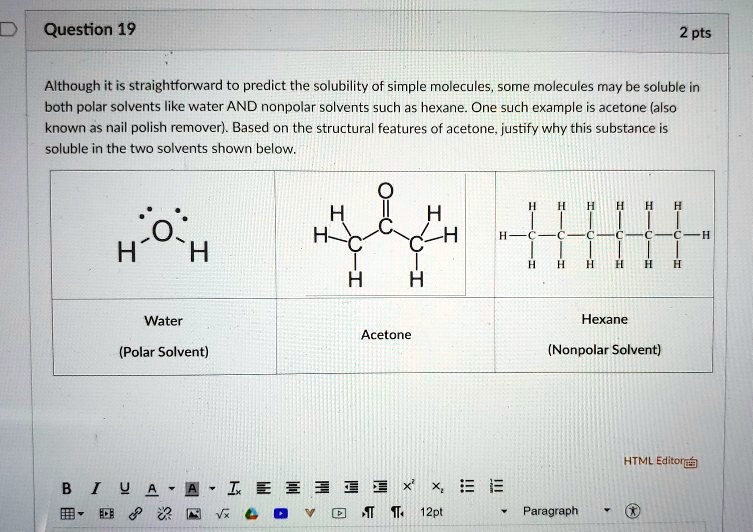
SOLVED: Although it is straightforward to predict the solubility of simple molecules, some molecules may be soluble in both polar solvents like water and nonpolar solvents such as hexane. One such example

Solubility of glucose, xylose, fructose and galactose in ionic liquids: Experimental and theoretical studies using a continuum solvation model - ScienceDirect

Acetone (propanone) is extremely soluble in water. Would you expect that 2- hexanone would be as soluble in water as acetone? Explain by comparing the chemical structures of the two compounds.

Solubility of Sugars and Sugar Alcohols in Ionic Liquids: Measurement and PC-SAFT Modeling | The Journal of Physical Chemistry B
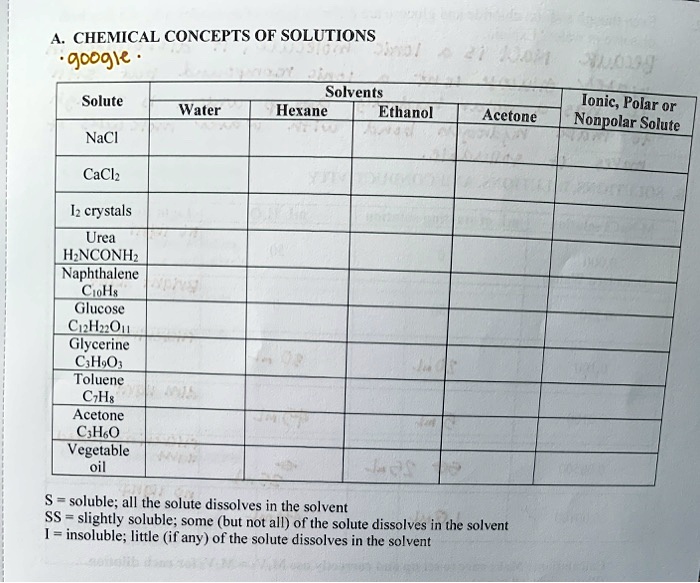
SOLVED: A. CHEMICAL CONCEPTS OF SOLUTIONS Solvents: Water, Hexane, Ethanol Solute: NaCl, Acetone, CaCl2, Urea, HNCONH2, Naphthalene, C6H5Cu, Glucose, CuI2, Glycerine, C6H12O6, Toluene, C6H6, Acetone, C6H5OH, Vegetable oil Solubility: - Soluble: all

Efficient One-Pot Production of 5-Hydroxymethylfurfural from Glucose in an Acetone–Water Solvent | Industrial & Engineering Chemistry Research

Solubility of d-Glucose in Water and Ethanol/Water Mixtures | Journal of Chemical & Engineering Data
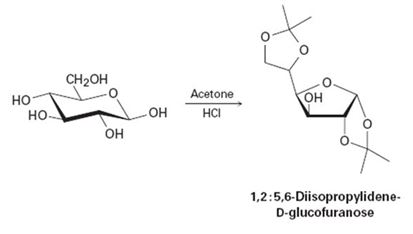
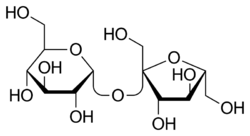
D-Glicose reacts with acetone in the presence of acid to yield the nonreducing 1, 2: 5, 6-diisopropylidene-D-glucofuranose. Propose a mechanism. | bartleby