SOLVED: The density of acetone is stated to be 791 kg/m^3, and 1 m^3 = 1*10^6 cm^3. what is the density of acetone in g/cm^3

An aqueous solution is prepared by diluting 3.30 mL of acetone density =0.789 g / mL to get the final volume of 75 mL. Find the mole fraction of acetone. Assume no

Calculate the volumes of acetone and ethanol that was used to mix together in order to produce 1 liter (1000 mL) of the equimolar mixture. Ethanol Mr = 46 and a density (

SOLVED: Acetone, a common solvent, has a density of 0.79 g/cm^3 at 20 degrees Celsius. What is the volume of 85.1 g of acetone at 20 degrees Celsius? What is the mass
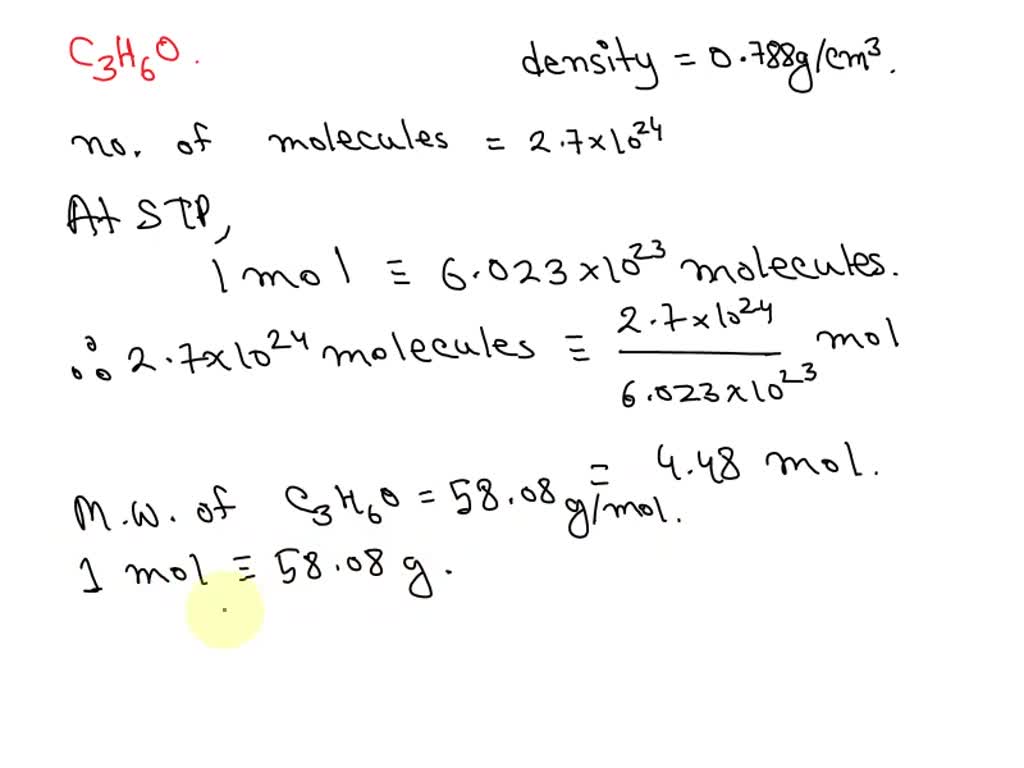
SOLVED: Acetone (C3H6O) has a density of 0.788 g/cm3. A sample of acetone contains 2.7×10^24 molecules of acetone. Determine the volume in mL of this sample.